10-15 minutes
Face and jaw shape
Facial sculpting
No downtime

Transform your appearance with our face slimming injections, the non-surgical solution to achieving that coveted V-shaped facial contour effortlessly.
Face slimming injections, or masseter injections, refer to treatments using botulinum toxin, such as Bo.tox, to contour and slim the face. By relaxing these muscles, botulinum toxin injections can reduce jaw size, leading to a more refined and aesthetically pleasing jawline.
This non-surgical procedure is for you if you are looking to achieve a softer, less angular facial appearance without the need for invasive surgery.
Facial slimming injections can effectively address several Cosmetic & Medical concerns, including:

Posted on Google
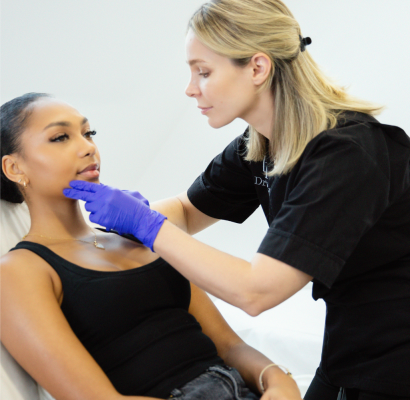
Choosing face slimming treatments over traditional procedures offers a less invasive, quicker, and more convenient option if you are looking to contour their face. Unlike surgical methods like liposuction or facelifts, non-surgical face slimming treatments—such as Bo.tox —require little to no downtime, allowing you to resume normal activities immediately. These treatments target specific areas, such as the jawline or cheeks, to reduce fat, tighten skin, or enhance bone structure without the need for cuts, stitches, or general anesthesia. Additionally, non-surgical options are more affordable, offer natural-looking results, and can be adjusted or repeated over time, providing flexibility and less risk compared to traditional surgical methods.

1. Consultation
The face slimming procedure at Dr. Ducu Clinics begins with a comprehensive consultation. This step ensures that the treatment is tailored to your specific needs and is safe for you. The consultation provides an opportunity to ask questions and understand the procedure, potential risks, and expected outcomes. The beautician will explain how the injections work, determine the appropriate dosage, and plan the injection sites to achieve the desired facial slimming effect.
2. Face slimming injections
During the injection procedure, the targeted areas are cleaned and marked. We will apply a topical Cosmetic & Medical to minimise discomfort. Using a fine needle, the practitioner injects a precise amount of dermal fillers (a combination of Bo.tox, Morpheus8 and Facetite) into the marked sites. The process is relatively quick, typically taking only a few minutes, and is performed with careful attention to detail to ensure symmetry and natural-looking results.
3. Aftercare
After receiving the injections, you will be advised to avoid rubbing or massaging the treated areas to prevent spreading the product to unintended muscles. You will be allowed to return to your daily activities immediately. However, it is recommended to avoid strenuous exercise, heat exposure, and lying down for a few hours post-treatment. You may experience slight swelling or bruising, which typically subsides within a few days.
4. Results
The results of face slimming injections start to become noticeable within a week, with full results typically seen in about two weeks as the muscles relax and reduce in size. The outcome is a softer, more contoured jawline that enhances the facial profile. The results can last from 3 to 6 months, and sometimes longer, depending on your individual response to the treatment. Repeat sessions may be necessary to maintain the slimming effect.
Choosing Dr. Ducu Clinics for facial slimming injections ensures you’re in the hands of experienced professionals who are specialised in advanced, non-surgical aesthetic treatments. Our team has extensive training and a proven track record of successful results, allowing you to feel confident that you’re receiving the highest standard of care.
We use only the latest, MHRA-approved techniques and products to ensure safety, effectiveness, and natural-looking results. Our personalised consultations ensure that we tailor the treatment to your unique facial structure and goals, giving you a customised approach. With a focus on professionalism, patient comfort, and long-term results, our clinic offers a trusted, welcoming environment where you can achieve the slimmer, more defined face you desire.
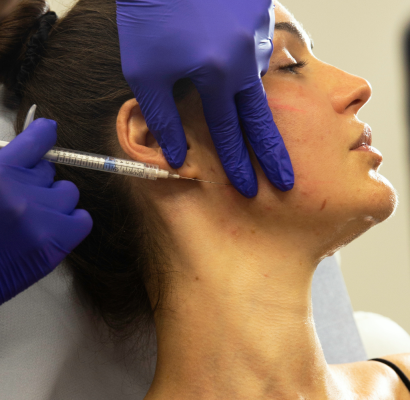

Non-surgical and minimally invasive: Facial slimming injections provide a non-surgical alternative to more invasive procedures.
Quick and convenient: The treatment is quick, often completed in just a few minutes, and the results are visible within a few weeks.
Customisable results: The amount and placement of injections can be tailored to your facial structure and desired outcomes.
Dual benefits: Beyond Cosmetic & Medical improvement, these injections can also reduce symptoms of bruxism (teeth grinding) and TMJ disorders, providing both cosmetic and therapeutic benefits.
Boosted self-confidence: Achieving a more Cosmetic & Medical pleasing facial contour can significantly enhance self-esteem and confidence.
Discover the transformative power of facial slimming injections, a single treatment packed with great benefits:
Ideal candidates for face slimming fillers are those seeking to soften a square or muscular jawline, often caused by genetics, bruxism, or habitual chewing, aiming for a slimmer, V-shaped facial profile. They should be healthy, with realistic expectations about the procedure.
Facial slimming injections use botulinum toxin, which works by temporarily relaxing the masseter muscles at the sides of the jaw. When these muscles are overactive or enlarged, they can give the face a square or widened appearance. By injecting botulinum toxin into these muscles, their activity is reduced, leading to a softer and more contoured jawline. This creates a slimmer facial profile as the muscle volume decreases and the excessive bulging of the jawline is minimised.
The effects of facial slimming injections typically start to become noticeable within a week to two weeks after the treatment. However, the full slimming effect usually becomes more evident after about a month, as it takes time for the masseter muscles to reduce in size and for the skin to contour to the new shape of the jawline.
The effects of a jawline slimming procedure using botulinum toxin injections typically last between 4 to 6 months. However, this can vary depending on your metabolism, the specific product used, and the dose administered. Over time and with repeated treatments, the results may last longer as the masseter muscles adapt to reduced activity levels.
Slight bruising and tenderness are normal with a filler injection, but 99% of the time there are no other problems. Most people really enjoy their filler results. It softens up after a few weeks and stays for a long time.
After the procedure the following activities are forbidden, for at least 3 weeks in most cases: manipulating the area(s) of injection, alcohol use, exposure to sun or other types of radiations, extreme temperatures (such as hot/cold showers, sauna, etc), using products for skin that are not recommended by the doctor for the area undergoing the procedure, makeup (may be not recommended for a period of time), any movements or actions of/on the area that can affect or traumatise the area (such as massaging the area, hitting, procedures or treatments on the area unless previously being assessed and deemed suitable by a healthcare practitioner).
There may be some swelling and bruising post procedure whichis a normal reaction, if any concerns please contact the Dr Ducu London team. We will always keep in contact with you to ensure you are well and for any enquires please feel free to contact us any time.
Chin injections are contraindicated in the following situations:
The price it’s determinate by the amount that the doctor will be using to achieve the results and the number of areas that need to be treated. The best course of action will be to book in a consultation with one of our doctors.
This procedure entails a combination of Bo.tox, Morpheus8 and Facetite. Is adapted to each one of our patients, depending on the person’s profile.
Apply a cool compress to the treatment area (do not apply ice directly to the skin) for 10 minutes every half an hour on the day of treatment. Avoid any heat inducing activities for 24 hours such as strenuous exercise, spas, saunas and hot showers.